Regulatory Compliance Timing in Drinkware Customization
Why Certification Assumptions Create Mid-Production Compliance Failures
In practice, this is often where customization decisions start to be misjudged. A procurement team receives a supplier's product catalog showing LFGB and FDA certifications for a standard stainless steel tumbler. The certificates are current, the test reports look comprehensive, and the supplier has clearly invested in compliance infrastructure. The buyer assumes that these certificates will cover their customized order—perhaps with a custom powder coating color, a silicone grip band, or a modified lid design. The purchase order is issued, the deposit is paid, and production begins. Three weeks later, the European distributor's compliance officer reviews the specifications and asks a simple question: "Do you have certification for this specific powder coating formulation?" The answer, more often than not, is no.
This scenario repeats itself across the corporate drinkware supply chain with surprising frequency. The fundamental misjudgment is treating regulatory compliance as a supplier attribute rather than a product-specific validation. When a factory holds an LFGB certificate for their standard product line, that certification applies to the exact material composition, surface finish, and component assembly tested by the accredited laboratory. The moment a buyer requests a customization—even something as seemingly minor as changing from matte black to glossy red powder coating—the certified configuration changes. The new coating may introduce different volatile organic compounds, use alternative curing agents, or contain pigments that weren't part of the original migration testing. From a regulatory standpoint, this is a different product, and it requires separate validation.
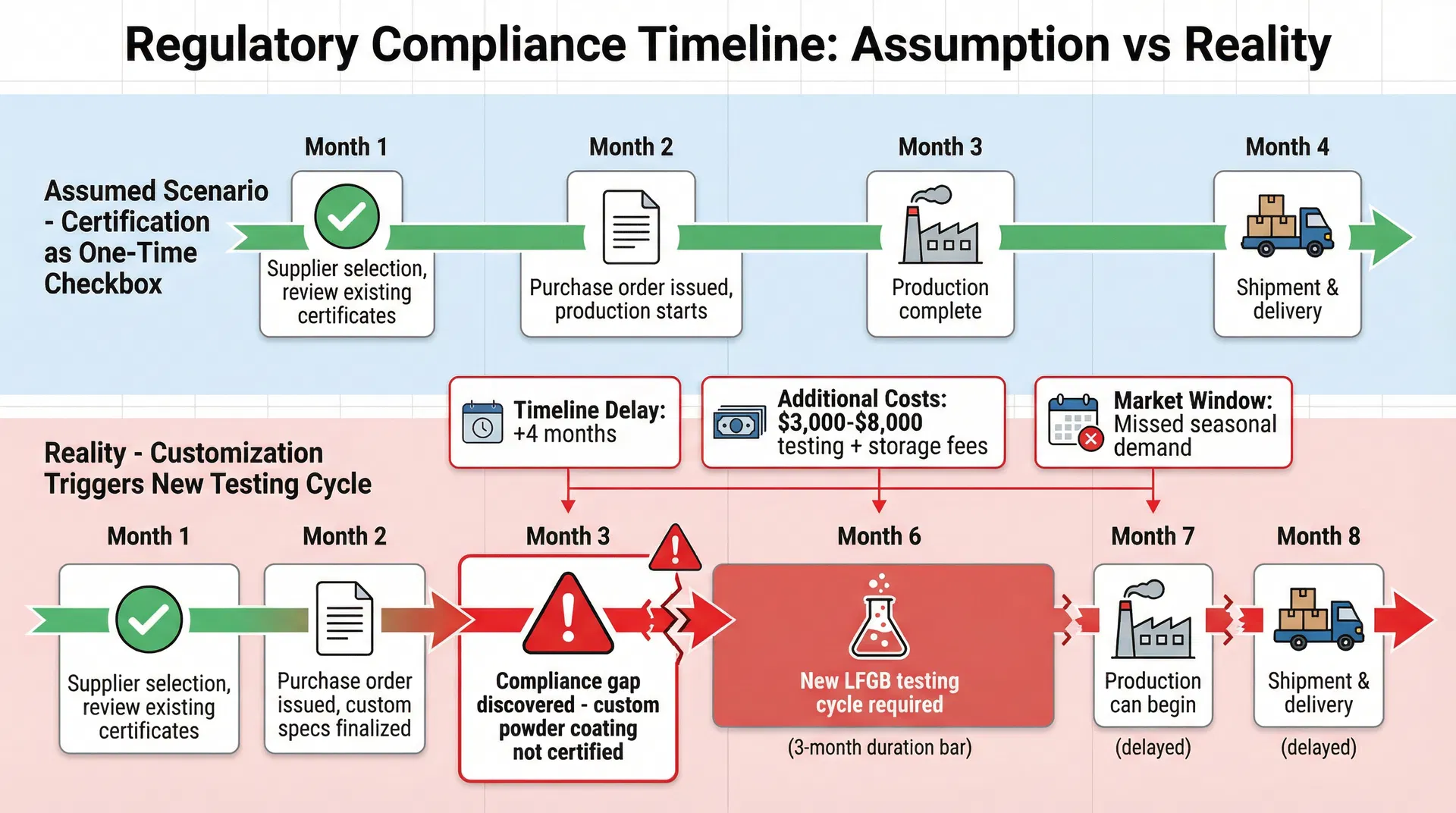
The timing implications of this misjudgment are severe. LFGB testing through an accredited German laboratory typically requires three months from sample submission to certificate issuance. This includes the laboratory's queue time, the 30-day migration testing period mandated by the standard, and the report compilation phase. If a compliance gap is discovered after production has started—or worse, after the first production batch is complete—the buyer faces an impossible choice. They can halt the order and wait three months for new testing, incurring storage costs and missing their market window. They can proceed without proper certification and risk customs rejection at the European border, which often results in the entire shipment being held or destroyed. Or they can attempt to renegotiate with the supplier to revert to a certified configuration, which may no longer be possible if custom tooling or materials have already been procured.
The cost mathematics of mid-production compliance failures are particularly unforgiving. Consider a 10,000-unit order of custom tumblers with a unit cost of $8.50. The total product value is $85,000. If the shipment is rejected at customs due to missing LFGB certification, the buyer not only loses the product value but also incurs air freight costs (typically $12,000-$18,000 for this volume), customs clearance fees, storage charges at the port, and potential destruction fees. The total loss can easily exceed $120,000. Even if the buyer chooses to halt production and wait for proper testing, they face three months of delayed revenue, potential contract penalties from their own customers, and the risk that seasonal demand windows will close. A compliance failure discovered in June means certification won't be available until September—well past the summer promotional season that may have been the entire rationale for the product launch.
What makes this misjudgment particularly insidious is that it often stems from reasonable assumptions. Buyers see that their supplier has invested in compliance infrastructure, maintains relationships with accredited testing laboratories, and can produce certificates for their standard product range. The supplier's sales team, eager to close the deal, may assure the buyer that "minor customizations" won't affect certification status. This assurance is rarely made with malicious intent; it often reflects the sales team's own lack of understanding about how material modifications interact with regulatory testing protocols. The factory's compliance officer, who would immediately recognize the issue, may not be consulted until production planning begins—by which point the commercial terms are locked and the timeline pressure is already building.
The regulatory framework itself contributes to the confusion. FDA compliance in the United States allows for a degree of self-declaration, particularly for materials that fall under established food contact substance regulations. A manufacturer can often demonstrate FDA compliance by showing that their materials match compositions already cleared through the Food Contact Notification process, without requiring product-specific third-party testing. LFGB, by contrast, mandates independent laboratory testing for each specific product configuration. A buyer accustomed to the more flexible FDA approach may not immediately grasp that the European standard operates under fundamentally different principles. When they request a custom color or surface finish, they don't realize they've just triggered a requirement for an entirely new testing cycle.
The component-level complexity of modern drinkware amplifies this risk. A typical insulated tumbler is not a single-material product. It consists of an inner stainless steel wall, an outer stainless steel wall, a vacuum seal, a powder-coated exterior finish, a plastic or silicone lid, a rubber gasket, and potentially a silicone grip band or base pad. Each of these components must individually comply with food contact regulations, and the assembled product must pass migration testing as a complete system. When a buyer requests a customization, they often focus on the most visible element—the exterior color or the logo placement—without recognizing that this change may affect multiple components. A switch from a standard black powder coating to a custom Pantone color may require a different coating supplier, which means different base resins, different curing agents, and different pigment formulations. Each of these changes introduces new substances that must be evaluated against LFGB's restricted substance lists and migration limits.

The timeline for discovering these issues follows a predictable pattern. Initial supplier selection occurs in Month 1, with the buyer reviewing the supplier's general capabilities and certifications. The purchase order is issued in Month 2, specifying custom colors and branding requirements. Production planning begins in Month 3, at which point the factory's technical team starts to source the specific materials needed for the customization. It is often at this stage—when the powder coating supplier is asked to provide material safety data sheets or when the lid manufacturer is asked about silicone formulation compliance—that the gaps become visible. By Month 3, the buyer has already committed to the supplier, communicated delivery timelines to their own customers, and potentially launched marketing campaigns around the product. The discovery that an additional three-month testing cycle is required doesn't just delay the project; it fundamentally undermines the entire business case.
The distinction between capacity and capability becomes critical in this context. A supplier may have the manufacturing capacity to produce 50,000 custom tumblers per month and the general capability to achieve LFGB certification for their standard products. But they may lack the specific capability to maintain certification across custom configurations, particularly if they source powder coatings or components from multiple sub-suppliers who don't themselves maintain comprehensive compliance documentation. When evaluating whether certain design choices will preserve certification validity, buyers need suppliers who understand the regulatory implications of material substitutions and who maintain direct relationships with accredited testing laboratories. This level of capability is not universal, even among suppliers who present themselves as compliance-ready.
The strategic response to this misjudgment requires shifting the compliance conversation to the earliest stages of the customization process. Before finalizing custom color selections, before approving artwork that requires specific printing methods, and certainly before issuing a purchase order, the buyer should explicitly ask: "Will this customization require new regulatory testing, and if so, what is the timeline and cost?" This question should be directed not to the sales team but to the supplier's compliance or technical department, with a request for written confirmation. If new testing is required, that three-month cycle must be built into the project timeline from the beginning, with clear milestones for sample submission, testing completion, and certificate receipt. The cost of testing—which can range from $3,000 to $8,000 per product configuration depending on the scope of required tests—should be explicitly included in the project budget and the commercial terms.
For buyers working across multiple markets, the compliance timing challenge multiplies. A product destined for both North American and European distribution may require parallel testing under both FDA and LFGB protocols, with different laboratories, different testing durations, and different documentation requirements. A powder coating formulation that passes FDA migration limits may fail LFGB's more stringent thresholds, requiring the buyer to either accept different product configurations for different markets or invest in reformulation work to achieve dual compliance. These decisions cannot be made mid-production; they must be resolved during the specification phase, before any tooling is created or materials are procured.
The relationship between customization complexity and certification risk is not linear. Adding a second custom color to a product line doesn't just double the testing requirement; it potentially creates an entirely new risk category if the two colors require different powder coating chemistries. Introducing a silicone component that wasn't part of the original certified design doesn't just add one more material to test; it introduces potential interactions between the silicone and the stainless steel, between the silicone and any beverages that will contact it, and between the silicone and the cleaning methods end users will employ. Each of these interactions must be evaluated through the lens of migration testing and sensory analysis, and each represents a potential point of failure that could invalidate the entire product's compliance status.
In the end, the misjudgment around regulatory compliance timing in drinkware customization stems from a fundamental category error. Buyers treat compliance as a binary attribute—either a supplier is compliant or they are not—when in reality, compliance is a product-specific state that must be actively maintained through every design decision and material selection. The supplier's existing certificates are valuable evidence of their capability to achieve compliance, but they are not transferable to customized configurations without explicit validation. Recognizing this distinction, and building the necessary testing timelines and costs into the project from the outset, is what separates procurement decisions that deliver on schedule from those that collapse under the weight of mid-production compliance failures.