Quality inspection in drinkware customization is frequently positioned as a final verification step that occurs after production is complete. This positioning creates a fundamental misunderstanding about what quality inspection actually accomplishes and when it should occur. The timing of quality inspection is not a logistical detail—it is a strategic decision that determines whether defects are caught and corrected at minimal cost, or whether they are released to the market where they become catastrophic liabilities. A procurement team that waits until production is complete to conduct quality inspection has already lost the opportunity to prevent the most expensive failures.
The distinction between pre-production sample inspection and post-production batch inspection represents two fundamentally different quality assurance strategies with dramatically different cost and risk profiles. Pre-production sample inspection occurs during the sample phase, before production tooling is finalized and before materials are committed to full-scale production. This inspection examines a small number of units (typically 3-5 samples) to verify that the design, materials, printing, and customization specifications are correct before the supplier commits to producing thousands of units. Post-production batch inspection occurs after production is complete, examining finished goods to verify that they meet specifications. The critical difference is that pre-production inspection can prevent production from beginning if defects are discovered, while post-production inspection can only identify defects after they have already been manufactured.
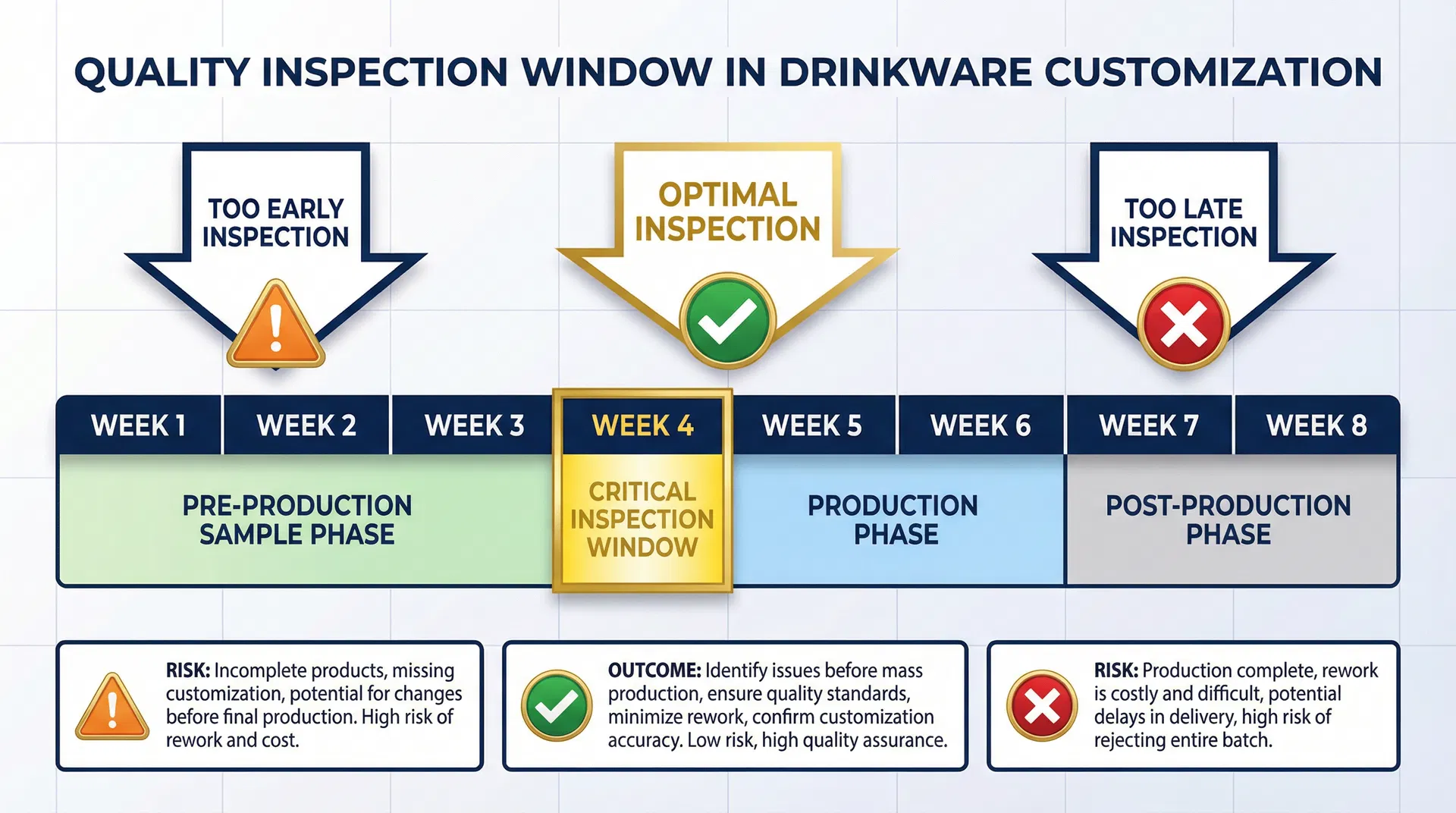
In practice, this is often where quality inspection timing decisions start to be misjudged—procurement teams assume that quality inspection is a binary choice between "inspect everything" or "inspect nothing," when the actual decision is about when to inspect and what to inspect at each phase. The pre-production sample phase is the critical inspection window because it is the last opportunity to verify that the supplier understands the specifications correctly before production begins. If a pre-production sample inspection reveals that the printing is off-color, the handle attachment is weak, or the customization is not meeting brand standards, the supplier can correct the issue before producing 10,000 units with the same defect. If the same defect is discovered during post-production inspection, the entire batch is already manufactured and the options are limited to expensive rework, accepting the defect, or rejecting the entire order.
The cost implications of inspection timing are dramatic and often underestimated. A pre-production sample inspection that costs $500-$2,000 can prevent a production run of 10,000 defective units, where each unit costs $5-$15 to manufacture. If a defect is discovered during pre-production inspection, the cost to correct it is typically $500-$5,000 (rework the sample, adjust tooling or processes, and produce a corrected sample). If the same defect is discovered after production is complete, the cost to address it is $50,000-$150,000 (rework 10,000 units, or scrap them entirely). The difference between catching a defect during pre-production inspection versus post-production inspection is a 10-30x cost multiplier. Yet many procurement teams avoid pre-production inspection because it adds 1-2 weeks to the timeline and costs a few thousand dollars, without fully recognizing that this decision creates a 50-100x cost risk if a defect is released to production.
The types of defects that are caught at different inspection phases determine the value of each inspection point. Pre-production sample inspection is most valuable for catching specification misunderstandings—the supplier interpreted the artwork incorrectly, chose the wrong material, or applied the wrong printing technique. These are defects that affect every unit in the production run and that cannot be fixed by reworking individual units. Post-production batch inspection is most valuable for catching manufacturing defects—a specific batch of material had a defect, a production run had a quality issue, or a small percentage of units failed to meet specifications. These defects typically affect only a portion of the batch and can sometimes be fixed by reworking individual units or by accepting a small number of defective units.
The decision about whether to conduct pre-production sample inspection is frequently driven by timeline pressure rather than by quality risk assessment. A procurement team that has a tight deadline may skip pre-production sample inspection to save 1-2 weeks, assuming that post-production inspection will catch any defects. This assumption is based on a fundamental misunderstanding of what post-production inspection can accomplish. If a pre-production sample inspection would have revealed that the supplier misunderstood the specification and produced 10,000 units incorrectly, post-production inspection will identify the defect, but it will not prevent the defect from being released to the market unless the procurement team is willing to reject the entire order and restart production. In most cases, the timeline pressure that motivated skipping pre-production inspection also prevents rejecting the entire post-production batch, so the defect is released to the market anyway.

The supplier's role in quality inspection timing creates additional complexity that procurement teams often fail to account for. A supplier that is accustomed to post-production inspection may not be prepared to conduct thorough pre-production sample inspection because they have not developed the processes or expertise to inspect samples comprehensively. A supplier that has never been asked to provide detailed sample inspection reports may not understand what information the procurement team needs to make a decision about whether to approve production. The procurement team must explicitly communicate what pre-production sample inspection should include—visual inspection of printing quality, color matching against approved standards, measurement of dimensions and tolerances, testing of functionality (lid seal, handle attachment, etc.), and verification of customization accuracy. Without this communication, the supplier may conduct a cursory inspection that misses critical defects.
The decision framework for pre-production sample inspection should be based on the complexity of the customization and the risk tolerance of the procurement team. For simple customizations (single-color logo printing on standard drinkware), pre-production sample inspection is still valuable but the risk of specification misunderstanding is lower. For complex customizations (multi-color artwork, custom colors, special printing techniques, or multiple customization elements), pre-production sample inspection is critical because the risk of specification misunderstanding is high and the cost of catching a defect during pre-production is dramatically lower than catching it after production. A procurement team should establish a clear decision rule: if the customization is complex or if the supplier is new or unfamiliar with the specific customization technique, pre-production sample inspection is mandatory. If the customization is simple and the supplier has a track record of quality, pre-production sample inspection can be optional but should still be considered.
The approval process for pre-production samples must be structured to ensure that approval decisions are made quickly and that the decision to approve production is based on clear quality criteria. A procurement team that receives a pre-production sample but does not have a clear process for evaluating it and making an approval decision will delay the project while the sample sits in a queue waiting for review. The approval process should specify who needs to review the sample (brand team, quality team, procurement team), what criteria they will use to evaluate it (does it match the approved artwork, is the color correct, are dimensions within tolerance, etc.), and how quickly they need to provide approval (typically within 2-3 business days). If the approval process is not structured, the timeline benefit of pre-production sample inspection is lost because the sample review becomes a bottleneck.
The contingency planning for pre-production sample inspection failures must be established before samples are produced. If a pre-production sample is rejected because it does not meet specifications, what is the process for correcting the issue and producing a revised sample? Can the supplier correct the issue and produce a revised sample within 1-2 weeks, or will the correction require significant rework that extends the timeline by 3-4 weeks? If the timeline cannot accommodate a revised sample, the procurement team faces a difficult choice: approve production with a known defect, or reject the order and restart with a different supplier. This choice should be made proactively during the planning phase, not reactively when a defective sample is received.
The documentation of pre-production sample inspection results provides valuable information for future projects and for managing supplier performance. Recording what was inspected, what defects were found, how they were corrected, and whether the corrected sample was approved creates a quality history that improves decision-making on future projects. A procurement team that has documented "supplier A consistently produces samples that require one revision before approval" will make different supplier selection decisions than a team that has no historical data. Without this documentation, the same quality issues recur across projects and the same inspection delays repeat indefinitely.